Long non-coding RNA-AK138945 regulates myocardial ischemia-reperfusion injury via the miR-1-GRP94 signaling pathway
doi: 10.2478/fzm-2024-0004
-
Abstract:
Objective Myocardial ischemia-reperfusion injury (MIRI) is one of the leading causes of death from cardiovascular disease in humans, especially in individuals exposed to cold environments. Long non-coding RNAs (lncRNAs) regulate MIRI through multiple mechanisms.This study explored the regulatory effect of lncRNA-AK138945 on myocardial ischemia-reperfusion injury and its mechanism. Methods In vivo, 8- to 12-weeks-old C57BL/6 male mice underwent ligation of the left anterior descending coronary artery for 50 minutes followed by reperfusion for 48 hours. In vitro, the primary cultured neonatal mouse ventricular cardiomyocytes (NMVCs) were treated with 100 μmol/L hydrogen peroxide (H2O2). The knockdown of lncRNA-AK138945 was evaluated to detect cardiomyocyte apoptosis, and a glucose-regulated, endoplasmic reticulum stress-related protein 94 (GRP94) inhibitor was used to detect myocardial injury. Results We found that the expression level of lncRNA-AK138945 was reduced in MIRI mouse heart tissue and H2O2-treated cardiomyocytes. Moreover, the proportion of apoptosis in cardiomyocytes increased after lncRNA-AK138945 was silenced. The expression level of Bcl2 protein was decreased, and the expression level of Bad, Caspase 9 and Caspase 3 protein was increased. Our further study found that miR-1a-3p is a direct target of lncRNA-AK138945, after lncRNA-AK138945 was silenced in cardiomyocytes, the expression level of miR-1a-3p was increased while the expression level of its downstream protein GRP94 was decreased. Interestingly, treatment with a GRP94 inhibitor (PU-WS13) intensified H2O2-induced cardiomyocyte apoptosis. After overexpression of FOXO3, the expression levels of lncRNA-AK138945 and GRP94 were increased, while the expression levels of miR-1a-3p were decreased. Conclusion LncRNA-AK138945 inhibits GRP94 expression by regulating miR-1a-3p, leading to cardiomyocyte apoptosis. The transcription factor Forkhead Box Protein O3 (FOXO3) participates in cardiomyocyte apoptosis induced by endoplasmic reticulum stress through up-regulation of lncRNA-AK138945. -
Key words:
- myocardial ischemia reperfusion /
- lncRNA /
- apoptosis /
- microRNA GRP94
-
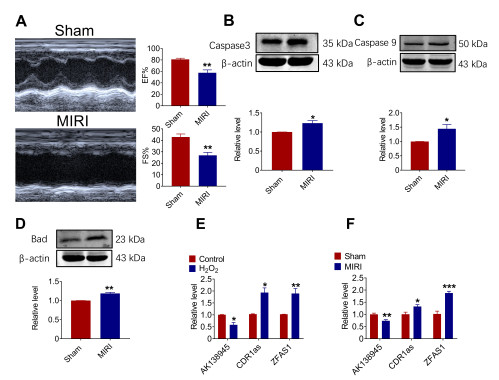
Figure 1. Expression of lncRNA-AK138945 in a mouse model of myocardial ischemia-reperfusion injury (MIRI). (A) Significant decreases in ejection fraction (EF) and fractional shortening (FS) in MIRI mice compared to the control group, indicating the successful establishment of the MIRI model. ** vs. Sham, P < 0.01, Sham, N = 6; MIRI, N = 4. (B-D) Western blot analysis on the expression levels of apoptotic proteins Caspase 9, Caspase 3, and Bad in myocardial tissue. * vs. Sham, P < 0.05; ** vs. Sham, P < 0.01, N = 3-5. (E) qPCR analysis on the expression of lncRNA-AK138945 in neonatal mouse cardiomyocytes after hydrogen peroxide treatment * vs. Control, P < 0.05; ** vs. Control, P < 0.01, N = 3. (F) Changes of lncRNA-AK138945 expression in cardiac tissue of MIRI model. * vs. Sham, P < 0.05; ** vs. Sham, P < 0.01; *** vs. Sham, P < 0.001, N = 3-4.
Figure 2. LncRNA-AK138945 knockdown facilitates cardiomyocyte apoptosis. (A) Verification of lncRNA-AK138945 knockdown efficiency by its siRNA (siAK138945)** vs. Control, P < 0.01; ### vs. +siAK138945, P < 0.001, N = 3. (B) Reduced cardiomyocyte viability by siAK138945, as measured by CCK8 assay. *** vs. Control, P < 0.001; ### vs. +siAK138945, P < 0.001, N = 6. (C) siAK138945 increased cardiomyocyte apoptosis by siAK138945 as unveiled by Tunel staining. ** vs. Control, P < 0.01; ## vs. +siAK138945, P < 0.01, N = 3. (D) siAK138945 increased cardiomyocyte death, as demonstrated by survival/death (Live/Dead) staining. Red fluorescence represents dead cells, and green fluorescence represents live cells. * vs. Control, P < 0.05; # vs. +siAK138945, P < 0.05, N = 3.
Figure 3. LncRNA-AK138945 knockdown anomaly alters the expression of apoptosis-related proteins in cardiomyocytes. (A-C) LncRNA-AK138945 knockdown by siAK138945 increases the expression levels of apoptotic proteins Caspase3, Caspase9, and Bad in cardiomyocytes. * vs. Control, P < 0.05; ** vs. Control, P < 0.01; *** vs. Control, P < 0.001; # vs. +siAK138945, P < 0.05; ## vs. +siAK138945, P < 0.01, N = 3-4. (D) siAK138945 significantly reduced the expression of Bcl2. *** vs. Control, P < 0.001; # vs. +siAK138945, P < 0.05, N = 5.
Figure 4. Experimental verification of miR-1a-3p as a downstream target of lncRNA-AK138945. (A) The predicted binding sites between miR-1a-3p and lncRNA-AK138945. (B) Luciferase reporter gene activity. *** vs. NC, P < 0.001, N = 3. (C) Upregulation of miR-1a-3p expression following lncRNA-AK138945 knockdown. * vs. +siNC, P < 0.05; ** vs. +siNC, P < 0.01, N = 3-6. (D-E) Downregulation of GRP94 expression post-lncRNA-AK138945 knockdown. ** vs. Control, P < 0.01; *** vs. Control, P < 0.001; # vs. +siAK138945, P < 0.05; ## vs. +siAK138945, P < 0.01, N = 5-6.
Figure 5. Inhibition of GRP94 induces cardiomyocyte apoptosis. (A-C) After treatment with a GRP94 inhibitor, the expression of apoptosis-related proteins Caspase3, Caspase9 and Bad was increased. *** vs. Control, P < 0.001; # vs. H2O2, P < 0.05, ## vs. H2O2, P < 0.01, N = 3-6. (D) The expression of anti-apoptotic protein Bcl2 was reduced after treatment with GRP94 inhibitor. ** vs. Control, P < 0.01; # vs. H2O2, P < 0.05, N = 6.
Figure 6. Effect of transcription factor FOXO3 on the AK138945-miR-1-GRP94 signaling pathway. (A) The expression of FOXO3 in myocardial tissue. *** vs. Sham, P < 0.001, Sham, N = 4; MIRI, N = 4. (B) Verification of FOXO3 overexpression efficiency.*** vs. Control, P < 0.001; # vs. FOXO3, P < 0.05, N = 4. (C-D) The expression levels of lncRNA-AK138945 and GRP94 are up-regulated after FOXO3 overexpression. * vs. Control, P < 0.05; *** vs. Control, P < 0.001; # vs. FOXO3, P < 0.05; ## vs. FOXO3, P < 0.01, N = 6. (E) FOXO3 overexpression downregulates the expression level of miR-1a-3p. ** vs. Control, P < 0.01; # vs. FOXO3, P < 0.05, N = 6.
-
[1] Clark D P, Hanke C W, Swanson N A. Dermal implants: safety of products injected for soft tissue augmentation. J Am Acad Dermatol, 1989(5 Pt 1); 21: 992-998. [2] Inoue T. Ischemia-reperfusion injury is still a big hurdle to overcome for treatment of acute myocardial infarction. J Cardiol, 2016; 67(4): 305-306. doi: 10.1016/j.jjcc.2015.09.002 [3] Poss J, Desch S, Eitel C, et al. Left ventricular thrombus formation after st-segment-elevation myocardial infarction: insights from a cardiac magnetic resonance multicenter study. Circ Cardiovasc Imaging, 2015; 8(10): e003417. doi: 10.1161/CIRCIMAGING.115.003417 [4] Lv L S, Zhou C L, Jin D H, et al. Impact of ambient temperature on life loss per death from cardiovascular diseases: a multicenter study in central China. Environ Sci Pollut Res Int, 2022; 29(11): 15791-15799. doi: 10.1007/s11356-021-16888-7 [5] Fonseca-Rodriguez O, Sheridan S C, Lundevaller E H, et al. Effect of extreme hot and cold weather on cause-specific hospitalizations in Sweden: A time series analysis. Environ Res, 2021; 193: 110535 doi: 10.1016/j.envres.2020.110535 [6] Hu J, Hou Z, Xu Y, et al. Life loss of cardiovascular diseases per death attributable to ambient temperature: A national time series analysis based on 364 locations in China. Sci Total Environ, 2021; 756: 142614. doi: 10.1016/j.scitotenv.2020.142614 [7] Xu E, Li Y N, Li T T, et al. Association between ambient temperature and ambulance dispatch: a systematic review and meta-analysis. Environ Sci Pollut Res Int, 2022; 29(44): 66335-66347. doi: 10.1007/s11356-022-20508-3 [8] Heo J B, Lee Y S, Sung S. Epigenetic regulation by long noncoding RNAs in plants. Chromosome Res, 2013; 21(6-7): 685-693. doi: 10.1007/s10577-013-9392-6 [9] Zhang Y, Jiao L, Sun L, et al. Lncrna Zfas1 as a Serca2a inhibitor to cause intracellular Ca(2+) overload and contractile dysfunction in a mouse model of myocardial infarction. Circ Res, 2018; 122(10): 1354-1368. doi: 10.1161/CIRCRESAHA.117.312117 [10] Sun F, Zhuang Y, Zhu H, et al. Lncrna Pcfl promotes cardiac fibrosis via mir-378/Grb2 pathway following myocardial infarction. J Mol Cell Cardiol, 2019; 133: 188-198. doi: 10.1016/j.yjmcc.2019.06.011 [11] Cai B, Zhang Y, Zhao Y, et al. Long noncoding RNA-DACH1 (dachshund homolog 1) regulates cardiac function by inhibiting SERCA2a (sarcoplasmic reticulum calciumATPase 2a). Hypertension, 2019; 74(4): 833-842. doi: 10.1161/HYPERTENSIONAHA.119.12998 [12] Su X, Lv L, Li Y, et al. Lncrna Mirf promotes cardiac apoptosis through the miR-26a-Bak1 axis. Mol Ther Nucleic Acids, 2020; 20: 841-850. doi: 10.1016/j.omtn.2020.05.002 [13] Sun L, Zhang Y, Zhuang Y, et al. Expression profile of long non-coding RNAs in a mouse model of cardiac hypertrophy. Int J Cardiol, 2014; 177(1): 73-75. doi: 10.1016/j.ijcard.2014.09.032 [14] Fonseca S G, Urano F, Burcin M, et al. Stress hyperactivation in the beta-cell. Islets, 2010; 2: 1-9. doi: 10.4161/isl.2.1.10456 [15] Zhang Y, Liu X, Zhang L, et al. Metformin protects against H2O2-induced cardiomyocyte injury by inhibiting the miR-1a-3p/GRP94 pathway. Mol Ther Nucleic Acids, 2018; 13: 189-197. doi: 10.1016/j.omtn.2018.09.001 [16] Liu X, Zhang Y, Du W, et al. MiR-223-3p as a novel microrna regulator of expression of voltage-gated K+ channel Kv4.2 in acute myocardial infarction. Cell Physiol Biochem, 2016; 39(1): 102-114. doi: 10.1159/000445609 [17] Martindale J J, Fernandez R, Thuerauf D, et al. Endoplasmic reticulum stress gene induction and protection from ischemia/reperfusion injury in the hearts of transgenic mice with a tamoxifen-regulated form of ATF6. Circ Res, 2006; 98(9): 1186-1193. doi: 10.1161/01.RES.0000220643.65941.8d [18] Tramentozzi E, Finotti P. Effects of purine-scaffold inhibitors on HUVECs: involvement of the purinergic pathway and interference with ATP. Implications for preventing the adverse effects of extracellular GRP94. Biochem Biophys Rep, 2019; 19: 100661. doi: 10.1016/j.bbrep.2019.100661 [19] Sengupta A, Molkentin J D, Paik J H, et al. Foxo transcription factors promote cardiomyocyte survival upon induction of oxidative stress. J Biol Chem, 2011; 286(9): 7468-7478. doi: 10.1074/jbc.M110.179242 [20] Zhan L F, Zhang Q, Zhao L, et al. LncRNA-6395 promotes myocardial ischemia-reperfusion injury in mice through increasing p53 pathway. Acta Pharmacol Sin, 2022; 43(6): 1383-1394. doi: 10.1038/s41401-021-00767-5 [21] Li M, Jiao L, Shao Y, et al. LncRNA-ZFAS1 promotes myocardial ischemia-reperfusion injury through DNA methylation-mediated Notch1 down-regulation in mice. JACC Basic Transl Sci, 2022; 7(9): 880-895. doi: 10.1016/j.jacbts.2022.06.004 [22] Ebner A, Poitz D M, Alexiou K, et al. Secretion of adiponectin from mouse aorta and its role in cold storage-induced vascular dysfunction. Basic Res Cardiol, 2013; 108(6): 390 doi: 10.1007/s00395-013-0390-9 [23] Xue Y, Yu X, Zhang X, et al. Protective effects of ginsenoside Rc against acute cold exposure-induced myocardial injury in rats. J Food Sci, 2021; 86(7): 3252-3264. doi: 10.1111/1750-3841.15757 [24] Cong P, Liu Y, Liu N, et al. Cold exposure induced oxidative stress and apoptosis in the myocardium by inhibiting the Nrf2-Keap1 signaling pathway. BMC Cardiovasc Disord, 2018; 18(1): 36. doi: 10.1186/s12872-018-0748-x [25] Park J, Kim S, Kim D H, et al. Whole-body cold tolerance in older Korean female divers "haenyeo" during cold air exposure: effects of repetitive cold exposure and aging. Int J Biometeorol, 2018; 62(4): 543-551. doi: 10.1007/s00484-017-1463-5 [26] Yin Z Q, Ding G B, Chen X, et al. Beclin1 haploinsufficiency rescues low ambient temperature-induced cardiac remodeling and contractile dysfunction through inhibition of ferroptosis and mitochondrial injury. Metabolism, 2020; 113: 154397. doi: 10.1016/j.metabol.2020.154397 [27] Jiang S, Guo R, Zhang Y, et al. Heavy metal scavenger metallothionein mitigates deep hypothermia-induced myocardial contractile anomalies: role of autophagy. Am J Physiol Endocrinol Metab, 2013; 304(1): E74-86. doi: 10.1152/ajpendo.00176.2012 [28] Zhang Y, Li L, Hua Y, et al. Cardiac-specific knockout of ET(A) receptor mitigates low ambient temperature-induced cardiac hypertrophy and contractile dysfunction. J Mol Cell Biol, 2012; 4(2): 97-107. doi: 10.1093/jmcb/mjs002 [29] Zhang Y M, Hu N, Hua Y A, et al. Cardiac over expression of metallothionein rescues cold exposure-induced myocardial contractile dysfunction through attenuation of cardiac fibrosis despite cardiomyocyte mechanical anomalies. Free Radic Biol Med, 2012; 53(2): 194-207. doi: 10.1016/j.freeradbiomed.2012.04.005 [30] Liang J, Yin K, Cao X, et al. Attenuation of low ambient temperature-induced myocardial hypertrophy by atorvastatin via promoting Bcl-2 expression. Cell Physiol Biochem, 2017; 41(1): 286-295. doi: 10.1159/000456111 [31] Yin K, Zhao L, Feng D, et al. Resveratrol attenuated low ambient temperature-induced myocardial hypertrophy via inhibiting cardiomyocyte apoptosis. Cell Physiol Biochem, 2015; 35(6): 2451-2462. doi: 10.1159/000374045 [32] Ebner A, Poitz D M, Alexiou K, et al. Secretion of adiponectin from mouse aorta and its role in cold storage-induced vascular dysfunction. Basic Res Cardiol, 2013; 108(6): 390. doi: 10.1007/s00395-013-0390-9 [33] Chen T, Gu Y, Bai G H, et al. MiR-1a-3p Inhibits apoptosis in fluoride-exposed LS8 cells by targeting Map3k1. Biol Trace Elem Res, 2023. [34] He R, Ding C, Yin P, et al. MiR-1a-3p mitigates isoproterenol-induced heart failure by enhancing the expression of mitochondrial ND1 and COX1. Exp Cell Res, 2019; 378(1): 87-97. doi: 10.1016/j.yexcr.2019.03.012 -
 fzm-4-1-31_ESM.pdf
fzm-4-1-31_ESM.pdf

-


 投稿系统
投稿系统


 下载:
下载: