Ethanol extract of cassia seed alleviates metabolic dysfunction-associated steatotic liver disease by acting on multiple lipid metabolism-related pathways
doi: 10.1515/fzm-2024-0017
-
Abstract:
Background and objective In northern China's cold regions, the prevalence of metabolic dysfunction-associated steatotic liver disease (MASLD) exceeds 50%, significantly higher than the national and global rates. MASLD is an important risk factor for cardiovascular and cerebrovascular diseases, including coronary heart disease, stroke, and tumors, with no specific therapeutic drugs currently available. The ethanol extract of cassia seed (CSEE) has shown promise in lowering blood lipids and improving hepatic steatosis, but its mechanism in treating MASLD remains underexplored. This study aims to investigate the therapeutic effects and mechanisms of CSEE. Methods MASLD models were established in male Wistar rats and golden hamsters using a high fat diet (HFD). CSEE (10, 50, 250 mg/kg) was administered via gavage for six weeks. Serum levels of total cholesterol (TC), triglyceride (TG), lowdensity lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), aspartate aminotransferase (AST), and alanine aminotransferase (ALT), as well as liver TC and TG, were measured using biochemical kits. Histopathological changes in the liver were evaluated using Oil Red O staining, Hematoxylin-eosin (H&E) staining, and transmission electron microscopy (TEM). HepG2 cell viability was assessed using the cell counting kit-8 (CCK8) and Calcein-AM/PI staining. Network pharmacology was used to analyze drug-disease targets, and western blotting was used to confirm these predictions. Results CSEE treatment significantly reduced serum levels of TC, TG, LDL-C, ALT, and AST, and improved liver weight, liver index, and hepatic lipid deposition in rats and golden hamsters. In addition, CSEE alleviated free fatty acid (FFA)-induced lipid deposition in HepG2 cells. Molecular biology experiments demonstrated that CSEE increased the protein levels of p-AMPK, p-ACC, PPARα, CPT1A, PI3K P110 and p-AKT, while decreasing the protein levels of SREBP1, FASN, C/EBPα, and PPARγ, thus improving hepatic lipid metabolism and reducing lipid deposition. The beneficial effects of CSEE were reversed by small molecule inhibitors of the signaling pathways in vitro. Conclusion CSEE improves liver lipid metabolism and reduces lipid droplet deposition in Wistar rats and golden hamsters with MASLD by activating hepatic AMPK, PPARα, and PI3K/AKT signaling pathways. -
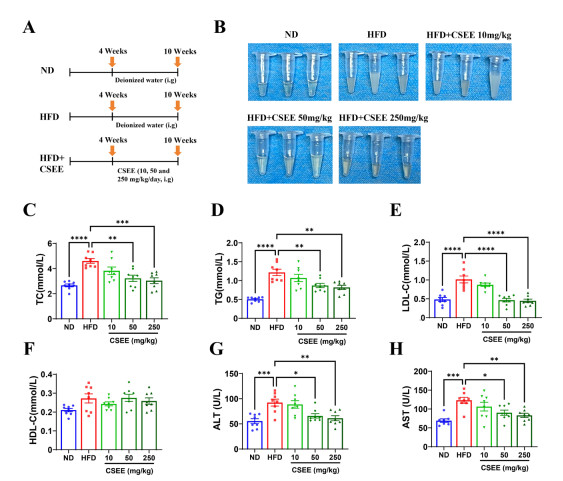
Figure 1. Cassia seed ethanol extract (CSEE) reduces elevated blood lipids and serum transaminase in rats fed with high-fat diet (HFD)
(A) Diagram illustrating the rat-feeding timeline. (B) Appearance of rat serum samples, N = 3. (C-H) Serum levels of (C) total cholesterol (TC), (D) triglyceride (TG), (E) lowdensity lipoprotein cholesterol (LDL-C), (F) high-density lipoprotein cholesterol (HDL-C), (G) alanine aminotransferase (ALT), and (H) aspartate aminotransferase (AST), measured in rats after CSEE oral administration for 6 weeks, N = 8 per group. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. ND: Normal Diet.
Figure 2. Cassia seed ethanol extract (CSEE) prevents high-fat-diet (HFD) feeding-induced hepatic morphological changes and lipid deposition in rats
(A) Representative images of rat livers from various groups. (B) Body weight, N = 8 per group. (C) Liver weight, N = 8 per group. (D) The weight ratio of liver and body, N = 8 per group. (E-F) Liver (E) total cholesterol (TC), and (F) triglyceride (TG) contents of rats from various groups, N = 8 per group. (G) Hematoxylin-eosin (H&E) staining of rat liver tissues. (H) Oil Red O staining of rat liver tissues. Scale bar: 100 μm; 50 μm, N = 8 per group. (I) Representative electron microscopic images of rat liver tissue slices from various groups. Scale bar: 5 μm. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. ND: Normal diet.
Figure 3. Cassia seed ethanol extract (CSEE) reduces elevated blood lipids and serum transaminase in hamsters fed with high fat diet (HFD)
(A) Diagram illustrating the feeding timeline for hamsters. (B) Appearance of hamster serum samples, N = 3. (C-H) Serum levels of (C) total cholesterol (TC), (D) triglyceride (TG), (E) low-density lipoprotein cholesterol (LDL-C), (F) high-density lipoprotein cholesterol (HDL-C), (G) alanine aminotransferase (ALT), and (H) aspartate aminotransferase (AST), measured in hamsters after CSEE oral administration for 6 weeks, N = 8 per group. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. ND: Normal diet.
Figure 4. Cassia seed ethanol extract (CSEE) prevents high-fat-diet (HFD) feeding-induced hepatic morphological changes and lipid deposition in hamsters
(A) Representative images of hamster livers from different groups. (B) Body weight, N = 8 per group. (C) Liver weight, N = 8 per group. (D) The weight ratio of liver and body, N = 8 per group. (E-F) Liver (E) total cholesterol (TC), and (F) triglyceride (TG) contents in hamsters of different groups, N = 8 per group. (G) Hematoxylin-eosin (H&E) staining of hamster liver tissues. (H) Oil Red O staining of hamster liver tissues. Scale bar: 100 μm; 50 μm, N = 8 per group. (I) Representative electron microscopic images of hamster livers of various groups. Scale bar: 2 μm. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. ND: Normal diet.
Figure 5. Cassia seed ethanol extract (CSEE) prevents free fatty acid (FFA)-induced increase in lipid content in HepG2 cells
(A) Representative images of Calcein acetoxymethyl ester/Propidium Iodide (Calcein-AM/PI) staining in HepG2 cells after CSEE treatment. Scale bar: 50 μm, N = 6 per group. (B) Cell viability of HepG2 measured by cell counting kit-8 (CCK8). N = 5 per group. (C-D) The contents of (C) total cholesterol (TC) and (D) triglyceride (TG) in HepG2 after CSEE treatment. N = 8 per group. (E) Representative images of Oil Red O staining in HepG2. N = 6 per group. (F) Representative images of Nile Red staining in HepG2 cells. N = 6 per group. **P < 0.01, ***P < 0.001, ****P < 0.0001. DMSO: Dimethyl sulfoxide.
Figure 6. Prediction and analysis of disease-drug targets
(A) Venn diagram of disease-related targets in five databases and GSE135251 dataset. (B) Volcano plot of differentially expressed genes (DEGs) in golden hamster livers between the high-fat-diet (HFD) group and the HFD + Cassia seed ethanol extract (CSEE) (250 mg/kg) group. (C) Venn diagram of DEGs. (D) Visualization of proteinprotein interaction (PPI) network analysis. (E) Gene ontology (GO) enrichment analysis of targets (Top 20 terms of biological process, molecular function and cellular component). (F) Kyoto encyclopedia of genes and genomes (KEGG) enrichment analysis of targets (Top 50 pathways). (G-I) Molecular docking analysis of CSEE and (G) Peroxisome proliferator-activated receptor α (PPARα), (H) fatty acid synthase (FASN), and (I) sterol regulatory element-binding protein 1 (SREBP1).
Figure 7. Cassia seed ethanol extract (CSEE) alleviates high-fat-diet (HFD)-induced hepatic steatosis by activating multiple signalling pathways
(A-B) Relative protein levels of the AMP-activated protein kinase (AMPK), peroxisome proliferator-activated receptor (PPAR), and phosphoinositide 3-kinase (PI3K)/AKT signalling pathways in (A) rat and (B) hamster livers, determined by Western blotting. N = 6 per group. (C-D) mRNA levels of the genes related to fatty acid synthesis, oxidation, uptake, and adipogenesis, as quantified by qRT-PCR. N = 8 per group. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. ND: Normal diet.
Figure 8. Inhibition of AMP-activated protein kinase (AMPK), peroxisome proliferator-activated receptorα (PPARα), and phosphoinositide 3-kinase (PI3K)/AKT reverses the beneficial effects of Cassia seed ethanol extract (CSEE) on lipid contents
(A-B) The contents of (A) total cholesterol (TC) and (B) triglyceride (TG) in HepG2 cells. N = 6 per group. (C) Representative images of Oil Red O staining in HepG2 cells. N = 6 per group. (D) Representative images of Nile Red staining in HepG2 cells. N = 6 per group. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. DMSO: Dimethyl sulfoxide. FFA: Free fatty acid.
-
[1] Man S, Deng Y, Ma Y, et al. Prevalence of liver steatosis and fibrosis in the general population and various high-risk populations: a nationwide study with 5.7 million adults in China. Gastroenterology, 2023; 165(4): 1025-1040. doi: 10.1053/j.gastro.2023.05.053 [2] Le M, Le D, Baez T, et al. Global incidence of non-alcoholic fatty liver disease: A systematic review and meta-analysis of 63 studies and 1, 201, 807 persons. J Hepatol, 2023; 79(2): 287-295. doi: 10.1016/j.jhep.2023.03.040 [3] Riazi K, Azhari H, Charette J, et al. The prevalence and incidence of NAFLD worldwide: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol, 2022; 7(9): 851-861. doi: 10.1016/S2468-1253(22)00165-0 [4] Qian Y, Che Z, Fu C, et al. Study on the association between dietary quality and overweight/obesity of Han nationality with cold in Yunnan plateau by DBI-16 - a study based on a multi-ethnic cohort in China. Diabetes Metab Syndr Obes, 2023; 16: 2311-2327. doi: 10.2147/DMSO.S420260 [5] Assy N, Kaita K, Mymin D, et al. Fatty infiltration of liver in hyperlipidemic patients. Dig Dis Sci, 2000; 45(10): 1929-1934. doi: 10.1023/A:1005661516165 [6] Li G, Peng Y, Chen Z, et al. Bidirectional association between hypertension and NAFLD: a systematic review and meta-analysis of observational studies. Int J Endocrinol, 2022; 2022: 8463640. [7] Ramanathan R, Patwa S, Ali A, et al. Thyroid hormone and mitochondrial dysfunction: therapeutic implications for metabolic dysfunction-associated steatotic liver disease (MASLD). Cells 2023; 12(24): 2806. doi: 10.3390/cells12242806 [8] Semmler G, Balcar L, Wernly S, et al. Insulin resistance and central obesity determine hepatic steatosis and explain cardiovascular risk in steatotic liver disease. Front Endocrinol (Lausanne), 2023; 14: 1244405. doi: 10.3389/fendo.2023.1244405 [9] Liu S, Liu Y, Wan B, et al. Association between vitamin D status and non-alcoholic fatty liver disease: a population-based study. J Nutr Sci Vitaminol (Tokyo), 2019; 65: 303-308. doi: 10.3177/jnsv.65.303 [10] Zhang J, Wang Q H, Miao B B, et al. Liver transcriptome analysis reveal the metabolic and apoptotic responses of Trachinotus ovatus under acute cold stress. Fish Shellfish Immunol, 2024; 148: 109476. doi: 10.1016/j.fsi.2024.109476 [11] Teng T, Sun G, Ding H et al. Characteristics of glucose and lipid metabolism and the interaction between gut microbiota and colonic mucosal immunity in pigs during cold exposure. J Anim Sci Biotechnol, 2023; 14(1): 84. doi: 10.1186/s40104-023-00886-5 [12] Haase C, Lopes S, Olsen A, et al. Weight loss and risk reduction of obesity-related outcomes in 0.5 million people: evidence from a UK primary care database. Int J Obes (Lond), 2021; 45(6): 1249-1258. doi: 10.1038/s41366-021-00788-4 [13] Sookoian S, Pirola C J. Resmetirom for treatment of MASH. Cell, 2024; 187(12): 2897-2897. e1. doi: 10.1016/j.cell.2024.05.009 [14] Chen Y, Chen X, Yang X, et al. Cassiae Semen: A comprehensive review of botany, traditional use, phytochemistry, pharmacology, toxicity, and quality control. J Ethnopharmacol, 2023; 306: 116199. doi: 10.1016/j.jep.2023.116199 [15] Ju M, Kim H, Choi J, et al. Cassiae semen, a seed of Cassia obtusifolia, has neuroprotective effects in Parkinson's disease models. Food Chem Toxicol, 2010; 48(8-9): 2037-2044. doi: 10.1016/j.fct.2010.05.002 [16] Kim S, Ban J, Kang H, et al. Anti-apoptotic effect of chrysophanol isolated from Cassia tora seed extract on blue-light-induced a2eloaded human retinal pigment epithelial cells. Int J Mol Sci, 2023; 24(7): 6676. doi: 10.3390/ijms24076676 [17] Patil U, Saraf S, Dixit V. Hypolipidemic activity of seeds of Cassia tora Linn. J Ethnopharmacol, 2004; 90(2-3): 249-252. doi: 10.1016/j.jep.2003.10.007 [18] Shih Y, Chen F, Wang L, et al. Discovery and study of novel antihypertensive peptides derived from Cassia obtusifolia seeds. J Agric Food Chem, 2019; 67(28): 7810-7820. doi: 10.1021/acs.jafc.9b01922 [19] Wang Q, Zhou J, Xiang Z, et al. Anti-diabetic and renoprotective effects of Cassiae Semen extract in the streptozotocin-induced diabetic rats. J Ethnopharmacol, 2019; 239: 111904. doi: 10.1016/j.jep.2019.111904 [20] Zhou F, Ding M, Gu Y, et al. Aurantio-obtusin attenuates nonalcoholic fatty liver disease through AMPK-mediated autophagy and fatty acid oxidation pathways. Front Pharmacol, 2021; 12: 826628. [21] Albillos A, de Gottardi A, Rescigno M. The gut-liver axis in liver disease: Pathophysiological basis for therapy. J Hepatol, 2020; 72(3): 558-577. doi: 10.1016/j.jhep.2019.10.003 [22] Luo H, Wu H, Wang L, et al. Hepatoprotective effects of Cassiae Semen on mice with non-alcoholic fatty liver disease based on gut microbiota. Commun Biol, 2021; 4(1): 1357. doi: 10.1038/s42003-021-02883-8 [23] Lei N, Song H, Zeng L, et al. Persistent lipid accumulation leads to persistent exacerbation of endoplasmic reticulum stress and inflammation in progressive NASH via the IRE1alpha/TRAF2 complex. Molecules, 2023; 28(7): 3185. doi: 10.3390/molecules28073185 [24] Li F, Jiang M, Ma M, et al. Anthelmintics nitazoxanide protects against experimental hyperlipidemia and hepatic steatosis in hamsters and mice. Acta Pharm Sin B, 2022; 12(3): 1322-1338. doi: 10.1016/j.apsb.2021.09.009 [25] Song S, Lai J, Wong G, et al. Can we use old NAFLD data under the new MASLD definition? J Hepatol, 2024; 80(2): e54-e56. doi: 10.1016/j.jhep.2023.07.021 [26] Eslam M, Newsome P, Sarin S, et al. A new definition for metabolic dysfunction-associated fatty liver disease: An international expert consensus statement. J Hepatol, 2020; 73(1): 202-209. doi: 10.1016/j.jhep.2020.03.039 [27] Liu J, Wang X, Zhu Y, et al. Theabrownin from dark tea ameliorates insulin resistance via attenuating oxidative stress and modulating IRS-1/PI3K/Akt pathway in HepG2 cells. Nutrients, 2023; 15(18): 3862. doi: 10.3390/nu15183862 [28] Dong Z, Shi H, Zhao M, et al. Loss of LCAT activity in the golden Syrian hamster elicits pro-atherogenic dyslipidemia and enhanced atherosclerosis. Metabolism, 2018; 83: 245-255. doi: 10.1016/j.metabol.2018.03.003 [29] Atteia H, AlFaris N, Alshammari G, et al. The hepatic antisteatosis effect of xanthohumol in high-fat diet-fed rats entails activation of AMPK as a possible protective mechanism. Foods, 2023; 12(23): 4214. doi: 10.3390/foods12234214 [30] Fang C, Pan J, Qu N, et al. The AMPK pathway in fatty liver disease. Front Physiol, 2022; 13: 970292. doi: 10.3389/fphys.2022.970292 [31] Steinberg G, Carling D. AMP-activated protein kinase: the current landscape for drug development. Nat Rev Drug Discov, 2019; 18(7): 527-551. doi: 10.1038/s41573-019-0019-2 [32] Katsuyama H, Yanai H, Adachi H, et al. A significant effect of pemafibrate on hepatic steatosis and fibrosis indexes in patients with hypertriglyceridemia. Gastroenterology Res, 2023; 16(4): 240-243. doi: 10.14740/gr1656 [33] Nakamura M, Yudell B, Loor J. Regulation of energy metabolism by long-chain fatty acids. Prog Lipid Res, 2014; 53: 124-144. doi: 10.1016/j.plipres.2013.12.001 [34] Yanai H, Yoshida H. Beneficial effects of adiponectin on glucose and lipid metabolism and atherosclerotic progression: mechanisms and perspectives. Int J Mol Sci, 2019; 20(5): 1190. doi: 10.3390/ijms20051190 [35] Habtemichael E, Li D, Camporez J, et al. Insulin-stimulated endoproteolytic TUG cleavage links energy expenditure with glucose uptake. Nat Metab, 2021; 3(3): 378-393. doi: 10.1038/s42255-021-00359-x [36] Leem Y, Bae J, Jeong H, et al. PRMT7 deficiency enhances adipogenesis through modulation of C/EBP-beta. Biochem Biophys Res Commun, 2019; 517(3): 484-490. doi: 10.1016/j.bbrc.2019.07.096 [37] Sopasakis V, Liu P, Suzuki R, et al. Specific roles of the p110alpha isoform of phosphatidylinsositol 3-kinase in hepatic insulin signaling and metabolic regulation. Cell Metab, 2010; 11(3): 220-230. doi: 10.1016/j.cmet.2010.02.002 [38] Tamilmani P, Sathibabu Uddandrao V, Chandrasekaran P, et al. Linalool attenuates lipid accumulation and oxidative stress in metabolic dysfunction-associated steatotic liver disease via Sirt1/Akt/PPRA-alpha/AMPK and Nrf-2/HO-1 signaling pathways. Clin Res Hepatol Gastroenterol, 2023; 47(10): 102231. doi: 10.1016/j.clinre.2023.102231 -


 投稿系统
投稿系统


 下载:
下载: